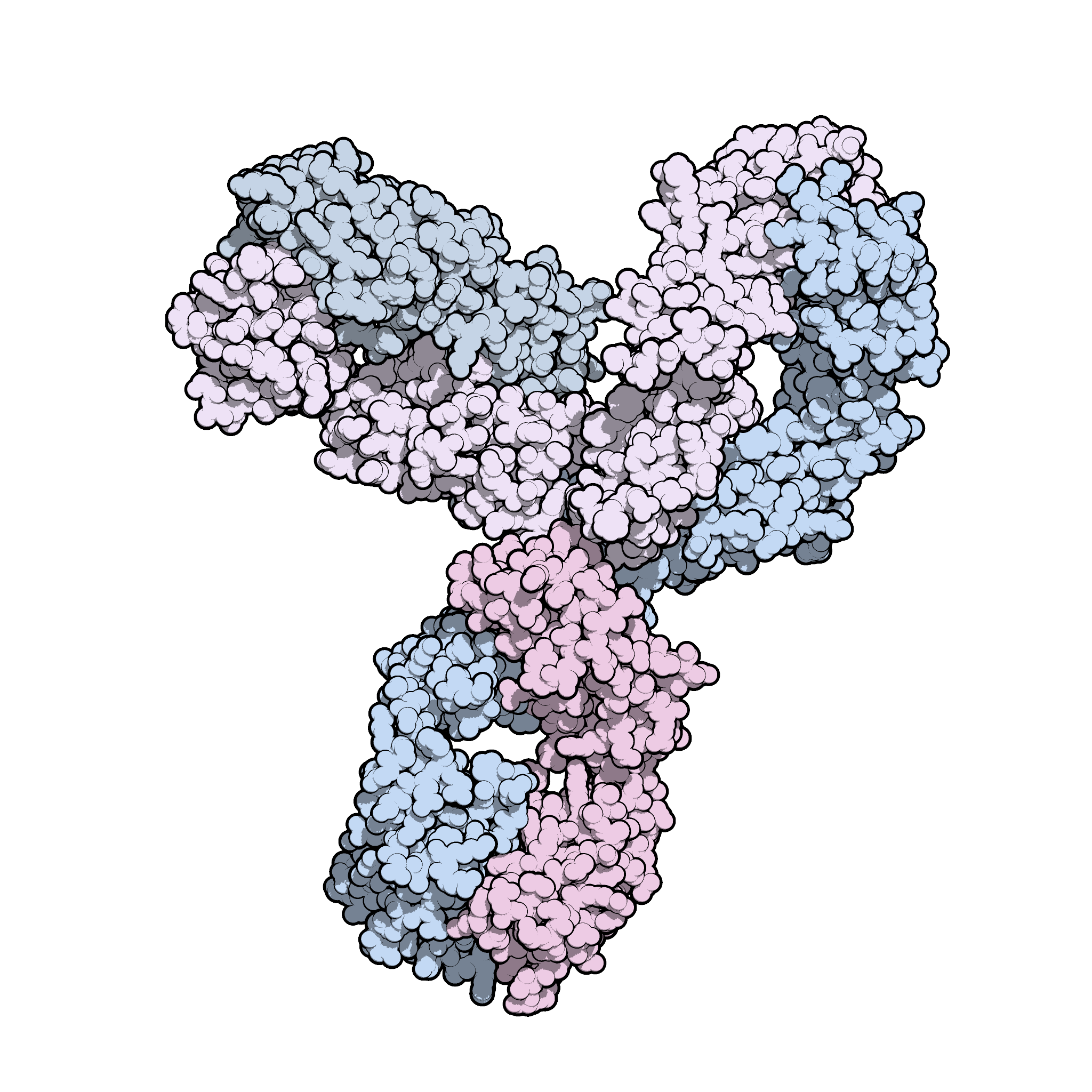
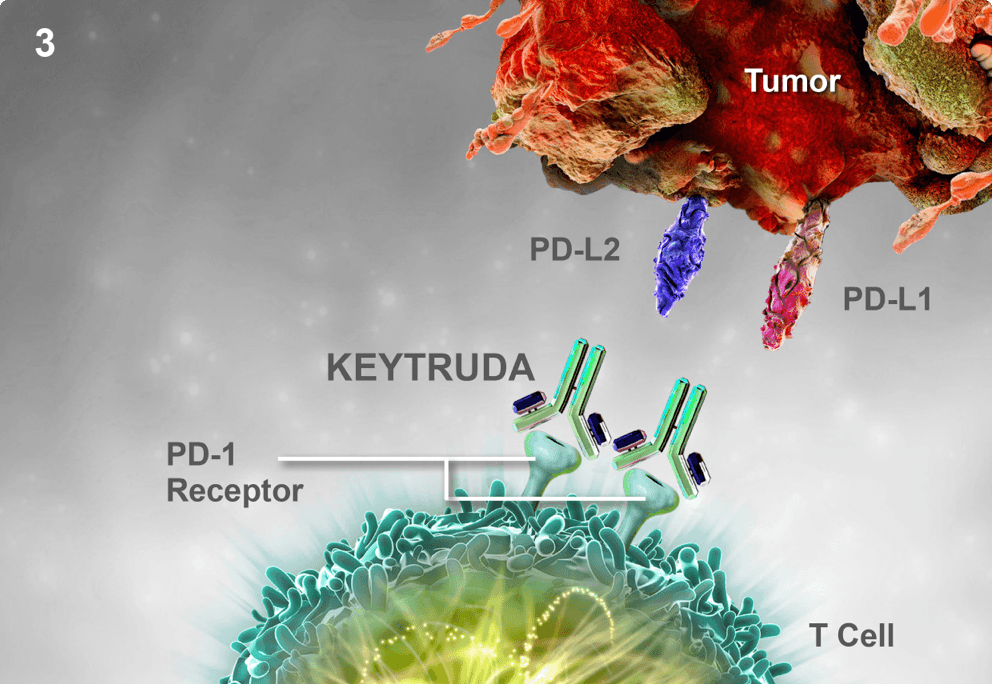
Keytruda® (pembrolizumab) — Drug Safety Updates • On June 19, 2015, the FDA approved updates to the Warnings and Precaution

Diabetic ketoacidosis following immunotherapy for lung cancer | Tidsskrift for Den norske legeforening

FDA Approves Merck's KEYTRUDA® (pembrolizumab) for Patients With MSI‑H/dMMR Advanced Endometrial Carcinoma, Who Have Disease Progression Following Prior Systemic Therapy in Any Setting and Are Not Candidates for Curative Surgery or Radiation
Merck posts early clinical data on subcutaneous Keytruda, setting stage for further studies | Fierce Pharma
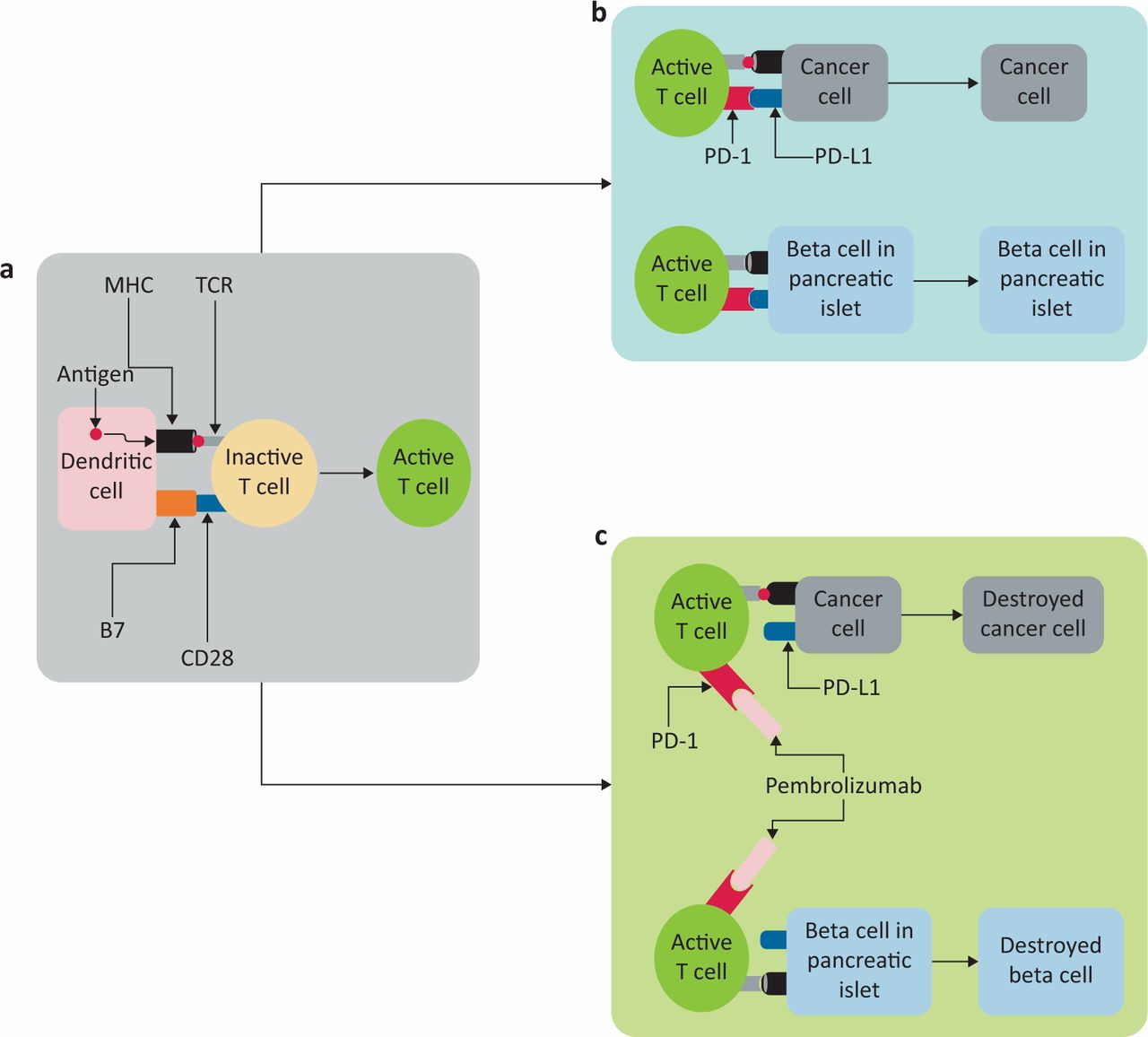
Is immune checkpoint inhibitor-associated diabetes the same as fulminant type 1 diabetes mellitus? | RCP Journals

Moderna und Merck geben bekannt, dass mRNA-4157/V940, ein personalisierter mRNA-Krebsimpfstoff, in Kombination mit KEYTRUDA(R) (Pembrolizumab) primären Endpunkt in Phase-2b-Studie KEYNOTE-942 erreicht hat

:max_bytes(150000):strip_icc()/VWH_Illustration_Drug_Keytruda-Pembrolizumab_Dennis-Madamba_Final-4a86ef9e12524d42aebbcd4126935dec.jpg)